Nature:
Anhydrous hydrogen fluoride is colorless, transparent liquid in the low-temperature. Boiling point is 19.4℃, melting point is -83.37℃. Critical temperature is 230.2℃; Critical pressure is 64 atm. Relative molecular weight is 20.01.The liquid density is 0.987 g/cm3(20℃);Gas density is 0.991g/L (0℃). It can easily volatilize to smokyfog under room and common temperature. Owing to its active chemical feature, it can react with alkaline, metal, oxide and silicate etc.
Under certain condition, it can be freely mixed with water into hydrofluoric acid with strong irritant smell, which will strongly corrode eyes, ear, nose and the membrane of throat. Moreover it seriously erodes human tooth and skeleton and calcifies them.
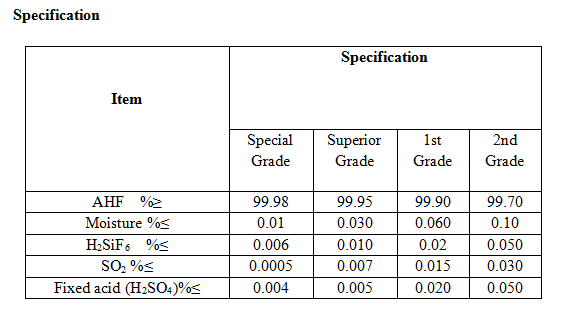
Standard
Anhydrous hydrogen fluoride conforms to stipulate of GB7746-1997.
Methods of analysis.Details of test methods can be supplied on request.
Packing: In 320kg steel cylinder or in 15 or 20MT tank or as clients’ requests. Filling ratio is needed as 0.832kg/L.
Storage and Transportation
Anhydrous hydrogen fluoride should be stored in draughty storeroom. Don’t shine for a long time and mix with inflammable and explosives. It should be treated as Grade A inorganic acid corrosive goods in transportation.
Safety Provision
When handle it, the protective clothing, gloves and safety goggles should be worn, or anhydrous hydrogen fluoride will infiltrate the skin quickly to harm your deep-seated body. The massive inspiration and scald may endanger personal safety. If contact the skin, rinse thoroughly with plenty of running water immediately, so that it can be diluted quickly.
Prevent heat, damp and rain during the transportation. When the shipment is made, please light load and unload preventing hit and inversion
1.It is a strong oxidant and sole raw material for making the element of fluorine
2.It is important main raw material for inorganic Fluoride and all kinds of organic fluoride.
3.The use of fluorosilicates: metal surface treatment agent, the oil recovery additive.
4.As etchant for glass and metal conditioner.
5.It is used to produce fluorine plastic and fluorine rubber.